AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
Back to Blog
Xe electron configuration8/5/2023  
The distribution of these unpaired electrons is called electron configuration and each type of arrangement has a name: Usually, atoms will have one or two unpaired electrons. It’s important to know where an electron might be found within an atom so as not to confuse it with another element when analyzing a compound or writing chemical formulasĮlectrons in an atom are distributed according to the electrons’ stability. If you don’t understand how they are distributed in atom, then understanding any other concept in this subject is difficult. Why is it important to know how electrons are distributed in an atom for chemistry purposes?Įlectrons are the backbone of all chemistry. As each new shell is filled, electrons will drop to the next lower energy level. The shells are occupied one level at a time first shell (K), second shell (L), third shell (M) etcetera until you reach nine where there is no further Shells. How do you read electron configuration?Įlectronic Configurations are determined by the number of electrons within an atom’s orbitals and their relative energy levels.What is the electronic configuration of 1 to 20 elements?.How do you write the electron configuration of an element?.What are the 3 principles of electron configuration?.Electronic configuration of elements in the periodic table.Hund’s Rule and filling of atomic orbitals.Pauli Exclusion Principle and filling of atomic orbitals. 
Aufbau principle and filling of atomic orbitals. 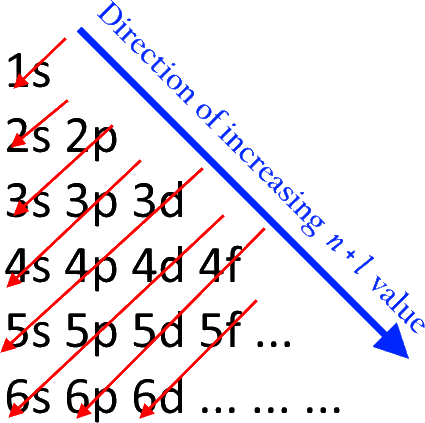
0 Comments
Read More
Leave a Reply. |
 RSS Feed
RSS Feed

